Abbott Research Team Home

Role title: Principal Investigator
Email: abbott@primate.wisc.edu
Another Lab HomepageAbbott Lab Research Focus
Polycystic Ovary Syndrome (PCOS) & Estrogen-Regulated Metabolism
- PCOS is the most common endocrine disorder impacting reproductive age women
- 10-18% prevalence of PCOS worldwide1
- Origin of PCOS still unknown
- 33% of PCOS cases accompanied with metabolic syndrome2
More specifically, our projects include:
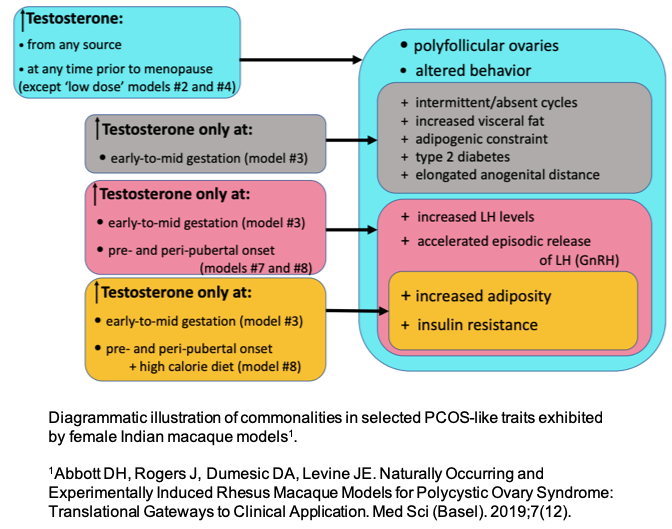
- Investigating how testosterone plays a role in PCOS etiology and presentation (Figure 1), such as, polycystic ovaries (Figure 2).
- Investigating the genetic basis of PCOS
- Exploring how estrogen regulates metabolism, cognitive function, and sexual behavior.
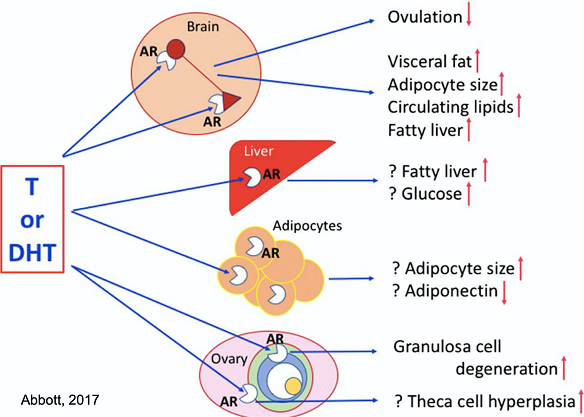
Figure 1. Schematic diagram of hypothetical sites for AR-mediated conversion to PCOS-like traits derived from the molecular modeling of Caldwell et al3. Question marks indicate deductive speculation based on Caldwell et al.
Possible molecular mechanisms generating the androgen excess still need to be further explored.
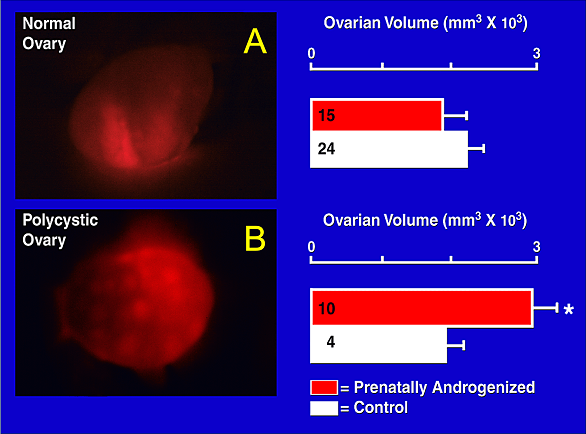
Figure 2 is representative of one of three diagnostic criteria for PCOS induced by heightened exposure to testosterone in utero. Figure 2A is an image of a normal ovary. Figure 2B is an image of a polycystic ovary.
Diagnostic Criteria for PCOS:
- Hyperandrogenism
- Intermittent/Absent Menstrual Cycles
- Polycystic Ovaries
- March WA, Moore VM, Willson KJ, Phillips DIW, Norman RJ, Davies MJ. The prevalence of polycystic ovary syndrome in a community sample assessed under contrasting diagnostic criteria. Hum Reprod 2010;25:544-51.
- Chandrasekaran, S, Sagili, H, Metabolic syndrome in women with polycystic ovary syndrome. The Obstetrician & Gynaecologist 2018; 20: 245-252. https://doi.org/10.1111/tog.12519
- Caldwell ASL, et al. (2017) Nueroendocrine androgen action is a key extraovarian mediator in the development of polycystic ovary syndrome. Proc Natl Acad Sci USA 114:E3334-E3343.
- Abbott, D. H. (2017). Neuronal androgen receptor: Molecular gateway to polycystic ovary syndrom? Proc Natl Acad Sci U S A, 114(16), 4045-4047. doi:10.1073/pnas.1703436114
Currently, there are two main areas of research focus: polycystic ovary syndrome (PCOS) afflicting up to 15% of women and female hyposexual dysfunction affecting ~10% of women. Both areas are collaborative efforts with PIs on campus and at other institutions.
In collaboration with Drs. Daniel Dumesic (UCLA), Jon Levine (Dept. of Neuroscience) and Dawn Davis (Dept. of Medicine), my lab developed two comprehensive nonhuman primate models for polycystic ovary syndrome (PCOS) that have been the vanguards for a multitude of animal and human studies aimed at determining developmental origins of this most common endocrinopathy in women. With NIH funding, we have translated insight gained from nonhuman primate models to discern pathophysiological mechanism causing discrete aspects of reproductive and metabolic dysfunction in women with PCOS, including abnormalities at the molecular level in the hypothalamus, adipose tissue, pancreas and ovary, as well as potential genetic contributions to naturally occurring PCOS.
Our most recent work identifies metabolic dysfunction and re-reprogramming of hypothalamic regulation as possibly the initial abnormalities in early developmental origins of PCOS. Most recently, we have been employing viral vector knockdown approaches to functionally identify molecular mechanisms. With regard to female hyposexual dysfunction, in collaboration with Drs. Alex Converse (Waisman Laboratory for Brain Imaging and Behavior), Yves Aubert (University of Bergen, Norway), Nicole Datson (BioMarin Nederland B.V., Netherlands) and Kelly Allers and Bernd Sommer (Boehringer Ingelheim, Germany), we have used MRI and PET neuroimaging, together with molecular neurobiology and digitized behavioral observations, to elucidate neural components contributing to female nonhuman primate social and sexual interactions with male pairmates. One of our recent studies contributed to a successful bench-to-bedside completion for flibanserin (Addyl), with the therapeutic achieving approval by the FDA for clinical use in treating hyposexuality in women, commencing October, 2015.
